현재 위치:홈 > 뉴스현황 > Our Events > Medicilon Instrument...
저자: 업로드:2017-05-09 조회수:
In recent years, the proportion of respiratory diseases gradually increased and the market prospects of inhalation administration are also growing by its virtue of the treatment of local or systemic diseases. In order to conform the market trends, Medicilon focus on inhalation of drug delivery services by enhancing the technical strength and improving service area and development. At the beginning of 2017, Medicilon purchased the inhalation exposure system equipment for both big and small animals. This newly bought system comply with OECD requirements which fulfill the single dose and multi-channel inhalation toxicity test requirements for rat, mice, dogs and other animals. (Pic 1-4.) With the help of the technical guidance and help by the engineers, Medicilon has successfully completed the instrument installation, staff training and 3Q verification. We are currently carrying out the pre-experiment and will soon be able to provide the reliable inhalation of drug delivery services to our clients.

Small animals (22 mice) with nasal inhalation exposure system
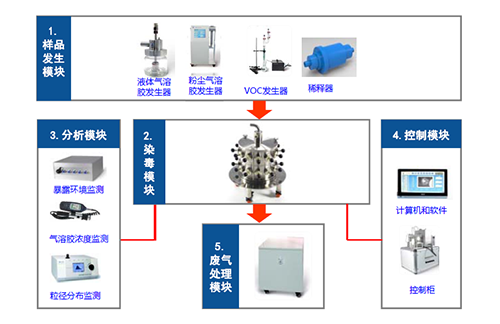
Four modules of nasal inhalation exposure system for small animals

Large animal (8 beagle dogs) with nasal inhalation exposure system
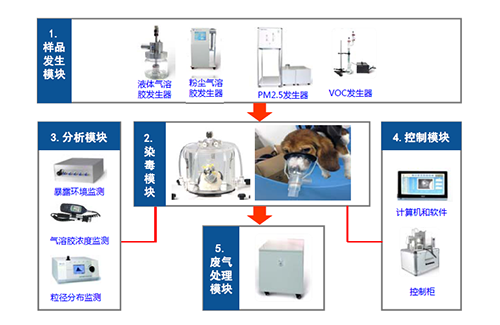
Four modules of nasal inhalation exposure system for large animals
Four modules of nasal inhalation exposure system for large animals
It is understood that the key factors that usually affect the results of inhalation exposure test are as follow:
1) Exposure to the environment (temperature, humidity, oxygen, carbon dioxide and ventilation)
2) Exposure characteristics of the test substance (concentration stability, homogeneity, t95 and MMAD
3) Inhalation dose (ventilation, sedimentation coefficient)
4) The physiological characteristics of animals (age, weight, gender, adaptability
Medicilon’s nasal inhalation exposure system is highly automated, enabling real-time monitoring and data traceability. For example, the aerosol particle distribution and concentration trend can be observed and adjusted by the aerosol mass concentration detection system. Our recent validation data showed that both the uniformity error and stability error of aerosol mass concentration comply with the OECD standard. The exposure control system could automatically control the aerosol concentration and pressure. This automated exposure process could reduce the risk of human infection. At the same time, the exposure data will be automatically saved to ensure the traceability of the data which fulfill the GLP requirement.